For this project we designed an outside class space that is comfortable and pleasant to use in all conditions. The classroom had to be comfortable and pleasant without the use of electricity. To start the project we learned about the Atomic state, structure, and heat. Next, we created a solar water heater to learn about conduction, convection, and radiation. Then, we created a survey to ask the student body and staff about what they wanted in their outdoor classroom. We took this information and used it to choose our site and what objects would be in our site. We tested materials to see what materials would be best for our site. We then made detailed blueprints that laid out our site plan and how to construct everything. We finally presented our sight to a panel of judges who graded not only our presentation, but also our features, material choice, blueprints, budget, model, and construable.
Solar Water Heater:
For this project we had to construct a solar water heater using: plastic heating, soft copper tubing, a water container, insulation, aluminum foil, cardboard, and a plastic sheet. We took a 24cm by 33cm by 8cm box and covered the inside with aluminum foil. The foil was used to reflect the Sun's rays on to the copper piping to heat up the wire. We used copper piping other than another metal piping because copper pipe is an amazing conductor. This means it transfers heat very well. This was important because we wanted the water to be heated as much as we could get it. We calculated the heat gained by using the equation Q = m C Delta T. (Q = M CAT) Our project was not the most successful. We noticed that more successful groups had more insulation and were shaped in a cone shape.
Client Need Survey:
For this part of the project we needed to figure out what our student body and staff wanted. To make it as easy as possible we created an online survey that could be accessed by anyone. We assigned one or two topics to each table group in our class. Each table group needed to create a question(s) about that topic. My table group was assigned materials as a topic. We created the question: What tools/materials would you need to make this space functional for you? The top two answers we received were wifi (by Students) and a whiteboard (by Staff). We used this information by making sure our site had wifi connection and making sure we added a whiteboard. The link below is a link to all the data we received from our survey.
https://docs.google.com/spreadsheets/d/1EQFHNU6X9jHaMICgCoxTiwbDdhbG9oIG7TqnHahVGLM/edit#gid=1781825641
Site Selection:
In order to start the blueprints for our project we needed to find a site. First, we walked around school and looked for three sites. The three sites we chose where behind the Spanish Building, near the Library, and next to the Art Building. The site next the Library was the highest rated, but we couldn't build there. Our next choice was behind the Spanish Building. This site was accessible by everyone, shaded by trees, and had wifi. We then took measurements of our site and started designing our plan.
Materials Testing Lab:
Our next job was to figure out what materials heated up the quickest and cooled down the quickest, or what materials absorbed the most water and what materials dried out the quickest. Each group in our class was assigned certain materials (ex. pine wood, cement, rubber, redwood, sand, etc.). We first put the materials under a heat lamp and recorded the temperature every five minutes for 30 minutes. Then we quickly put the materials in the refrigerator and recorded the temperature every five minutes. For the water part of the lab we weighed our material and wrote down the weight. We then submerged the material completely under water for 10 minutes. After the ten minutes were up, we took the materials out of the water and weighed them again. If a material weighed more then it did when it was dry we knew it absorbed more water. Lastly, we put our materials on the counter and let them sit for 10 more minutes. Once the 10 minutes were up we weighed them again to see h0w much they dried. The link below is a link to all of our recorded data.
https://docs.google.com/spreadsheets/d/1ddypjn9ikkl8zxYncXGBVuCyJaUOLF9J8zEToDIvsR4/edit#gid=0
Presentation:
On March 14, 2016, our class presented our building designs to a panel of judges. The judges graded us on our presentation tactics, our key features, usability, client need, energy location, material choice, blueprints, budget, model, and feasibility. Some of my group's key features were a wheel chair ramp and a whiteboard. Our site was usable because it is close to the campus and accessible to everyone. We made sure our site had a whiteboard and wifi for the client need. Our site was well shaded for energy location. We used pressure treated wood for anything that touched the ground and found many free items for material choice. We were the only group to give the judges a packet of our blue prints for the blueprint grade. We were well under budget and and had a full material list with all the prices (link below). We also added a 3-D scale model of our table for the judges.
https://docs.google.com/spreadsheets/d/1YA9mvM5Ev2WTOY2M7mQEaYizXc6uMRtogIa3cXa8WA/edit#gid=0
Wind Mill Lab:
Our objective for this lab was to design a wind turbine blade to maximize voltage output at low, medium, and high wind speeds for VAWT and HAWT. My group chose to work exclusively on VAWT's. We discovered under low wind conditions the spiral design was the best VAWT we created. Our goal was to maximize voltage using low wind settings and paper wings. Under the lowest wind condition our spiral design got 0.18 volts. The closest design was the tri-6 which created 0.16 volts. At the second lowest setting the spiral design got 0.4 volts. The closest to that was the tri-quad at 0.25 volts. We think the spiral design worked as well as it did for two reasons. First, the large blades. These blades helped because they trapped the most air. Next, the number of blades helped. This helped because the air was never pushing on the backs of other blades at the same time it was pushing on the front too. Clearly , the spiral design was the best under low wind conditions for VAWT's.
Our objective for this lab was to design a wind turbine blade to maximize voltage output at low, medium, and high wind speeds for VAWT and HAWT. My group chose to work exclusively on VAWT's. We discovered under low wind conditions the spiral design was the best VAWT we created. Our goal was to maximize voltage using low wind settings and paper wings. Under the lowest wind condition our spiral design got 0.18 volts. The closest design was the tri-6 which created 0.16 volts. At the second lowest setting the spiral design got 0.4 volts. The closest to that was the tri-quad at 0.25 volts. We think the spiral design worked as well as it did for two reasons. First, the large blades. These blades helped because they trapped the most air. Next, the number of blades helped. This helped because the air was never pushing on the backs of other blades at the same time it was pushing on the front too. Clearly , the spiral design was the best under low wind conditions for VAWT's.
Concepts:
Nucleus - center of an atom
Molecule- multiple atoms bonded together (ex: H2O, CO2, C6H12O6)
Compound -type of molecule with different types of elements bonded together
Isotope -an atom with an unusual number of neutrons
States of Matter -Liquid - molecules are free flowing and density of molecules are denser than gas but less dense then solid
-Solid - molecules hold a shape and the most dense
-Gas - molecules are free flowing and least dense
Conduction -transfer of heat through a solid
Convection - transfer of heat through fluid (liquid/gas)
Heat - a form of thermal energy (unit: J)
Thermal Conductivity - the rate at which material transfers heat
Thermal Resistance - the ability of material to resist transferring heat
Archimede's principle - an immersed object is buoyed up by a force equal to the
weight it displaces
Bernoulli's Principle - at speed of a fluid increases the pressure decreases
Specific Heat Capacity - constant for a substance that measures how slowly it heats up
High Specific Heat - warms up slowly, holds lots of heat well
Low Specific Heat - warms up quickly, doesn't hold heat well
Water - has a high specific heat capacity of 1cal/GdegreesC or 4.184J/GdegreesC
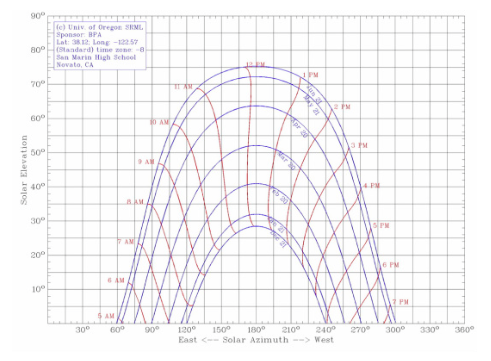
Sun Angles - Below is a picture of the Sun's Angles throughout the year. We used this chart to make sure our site has shade not only in the winter, but also in the summer, fall, and spring. It also helped us make sure our solar water heater got the most amount of sun possible.
Nucleus - center of an atom
Molecule- multiple atoms bonded together (ex: H2O, CO2, C6H12O6)
Compound -type of molecule with different types of elements bonded together
Isotope -an atom with an unusual number of neutrons
States of Matter -Liquid - molecules are free flowing and density of molecules are denser than gas but less dense then solid
-Solid - molecules hold a shape and the most dense
-Gas - molecules are free flowing and least dense
Conduction -transfer of heat through a solid
Convection - transfer of heat through fluid (liquid/gas)
Heat - a form of thermal energy (unit: J)
Thermal Conductivity - the rate at which material transfers heat
Thermal Resistance - the ability of material to resist transferring heat
Archimede's principle - an immersed object is buoyed up by a force equal to the
weight it displaces
Bernoulli's Principle - at speed of a fluid increases the pressure decreases
Specific Heat Capacity - constant for a substance that measures how slowly it heats up
High Specific Heat - warms up slowly, holds lots of heat well
Low Specific Heat - warms up quickly, doesn't hold heat well
Water - has a high specific heat capacity of 1cal/GdegreesC or 4.184J/GdegreesC
Sun Angles - Below is a picture of the Sun's Angles throughout the year. We used this chart to make sure our site has shade not only in the winter, but also in the summer, fall, and spring. It also helped us make sure our solar water heater got the most amount of sun possible.
Law of Thermodynamics:
Zero Law - explains temperature - if two systems are in thermal equilibrium with a third system. They are all in equilibrium. Ex. A=B B=C A must equal C
First Law - Conservation of Energy - energy is neither created or destroyed, but transferred from one form to the next form. Heat is a form of energy.
Second Law - Entropy increases - disorder increases, over time everything becomes the same energy - Temperature also follows this law.
Third Law - Temperature can never get to absolute zero, heat always exists.
Justification of Energy:
For this part of the project we learned about different energy sources (pros, cons, and how they work) and how it is affecting our world we live in. Below is the link to all of the notes we took.
https://docs.google.com/document/d/1xNlfcVQ0EfdhCoEf6vTE0RzP6w0dYkSAyULe8hU66TA/edit
REFLECTION:
During this project I learned a few things about myself. First, I learned that I can get off task easily when times get boring. A way for me to stay on task is two take a quick walk outside so I can come in the room ready to focus. Another thing I learned is I like to be the leader all the time. This can be good, but also can be bad because other people need to be the leader too. Two things I improved on are keeping my group on task most of the time. I did this by letting my group have a little bit of time each day to have a break. Another thing I improved on was taking ideas from everyone in my group. This helped many times during our project. It helped during the making of the table and the platform. In the end, I loved this project and I hope when we actually build the site it will be as amazing as I picture it to be.
Zero Law - explains temperature - if two systems are in thermal equilibrium with a third system. They are all in equilibrium. Ex. A=B B=C A must equal C
First Law - Conservation of Energy - energy is neither created or destroyed, but transferred from one form to the next form. Heat is a form of energy.
Second Law - Entropy increases - disorder increases, over time everything becomes the same energy - Temperature also follows this law.
Third Law - Temperature can never get to absolute zero, heat always exists.
Justification of Energy:
For this part of the project we learned about different energy sources (pros, cons, and how they work) and how it is affecting our world we live in. Below is the link to all of the notes we took.
https://docs.google.com/document/d/1xNlfcVQ0EfdhCoEf6vTE0RzP6w0dYkSAyULe8hU66TA/edit
REFLECTION:
During this project I learned a few things about myself. First, I learned that I can get off task easily when times get boring. A way for me to stay on task is two take a quick walk outside so I can come in the room ready to focus. Another thing I learned is I like to be the leader all the time. This can be good, but also can be bad because other people need to be the leader too. Two things I improved on are keeping my group on task most of the time. I did this by letting my group have a little bit of time each day to have a break. Another thing I improved on was taking ideas from everyone in my group. This helped many times during our project. It helped during the making of the table and the platform. In the end, I loved this project and I hope when we actually build the site it will be as amazing as I picture it to be.